

Image: Polar stratospheric clouds over Kiruna, Sweden, taken by Saga Dagnesjö, Idivuoma fam Blind, and Sheila Kirkwood.
Atmospheric chemistry
Our atmosphere is made of 78.08% N2, 20.95% O2, and 0.93% Argon. The remaining ~0.40% is made up of gases such as water vapor, CO2, organic compounds, and free radicals. But this is where all the action is! Free radicals have an odd number of electrons; they react with just about everything because they go around trying to pair off that odd electron.
How are free radicals generated in the first place? They form when molecules absorb light. Sometimes, the molecule absorbs ultraviolet light with enough energy to break a chemical bond; when this happens, it will generate two free radicals, one from each piece:
A-B + UV light → A• + B•Initiation
A• + A-B → B• + A-APropagation
B• + A-B → A• + B-B
A• + B• → A-BTermination
When a free radical reacts with another species, it typically generates another free radical because it steals just one electron. This propagates a chain reaction that continues until two radicals find each other to terminate that cycle. These free-radical chain reactions drive atmospheric chemistry at the surface and into the upper atmosphere; they are the reason we have smog in Los Angeles, but they are also the reason we have an ozone "shield" in the stratosphere protecting us from harmful UV radiation.
I study the kinetics and mechanisms of these free-radical reactions in the laboratory, and I examine their atmospheric implications. Understanding how these reactions progress in the lower atmosphere (the troposphere) can lead to more effective air-quality regulations and public policy, whereas understanding these reactions in the upper atmosphere (the stratosphere) can give us ways of tracing how air moves around the atmosphere.
- Key Findings:
- Alkynes (released from automobile emissions and coal burning) react differently from alkanes and alkenes in the atmosphere. Instead of forming peroxy radicals that can increase ground-level ozone in urban areas, they form stable diacetyls when oxidized in the atmosphere. As such, they contribute more to particle formation in urban areas. Link to JPCA article
- Stratospheric 16O13C18O is enriched relative to tropospheric 16O13C18O. This is especially pronounced in the the wintertime stratospheric polar vortex, where intruding mesospheric air and/or CO2 isotope-exchange reactions on polar stratospheric clouds generate the largest 16O13C18O enrichments ever observed in nature. Link to PNAS article
- The covariation of stratospheric 12C16O2, 16O12C18O, 16O12C17O, and 16O13C18O is a good tracer for O(1D) in the stratosphere. By comparing stratospheric distributions with laboratory experiments, the integrated effective isotopic composition of O(1D) in the midlatitude stratosphere was determined. It is a valuable constraint on the photochemistry of the ozone layer. Link to PNAS article
- The covariation of 16O16O, 16O17O, 16O18O, 17O18O, and 18O18O traces O(3P) concentrations, the atmospheric temperature profile, and stratosphere-troposphere exchange. While we're still working out the details of how these species covary in the different parts of the atmosphere, they are a promising new tracer of gas-phase chemistry and other atmospheric properties. Pilot studies of the isotopic composition of O2 in bubbles trapped in glacial ice are underway, with the goal of understanding variations in gas-phase chemistry and atmospheric dynamics over the last 60,000 years. Link to JGR article
- Alkynes (released from automobile emissions and coal burning) react differently from alkanes and alkenes in the atmosphere. Instead of forming peroxy radicals that can increase ground-level ozone in urban areas, they form stable diacetyls when oxidized in the atmosphere. As such, they contribute more to particle formation in urban areas. Link to JPCA article
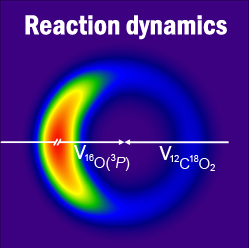
Image: Experimental velocity flux diagram for the oxygen isotope exchange reaction O(3P) + CO2.
Chemical reaction dynamics at high collision energies
When atoms and molecules react, what happens internally? How different is the energy distribution before and after a collision? Is reactivity inhibited or enhanced when you change the amount of available energy and how that energy is distributed? These are the fundamental questions we ask in chemical reaction dynamics.
At high collision energies, reaction pathways inaccessible under milder conditions become important. These reactions are not very common at Earth's surface, but high in the atmosphere (e.g., the thermosphere, 100-1000 km altitude), temperatures can reach 2000-3000°C! Determining which reactions and mechanisms dominate under these conditions is challenging, however, because most computational methods work best at low energies. Dynamics experiments, however, can test these methods.
I study the dynamics of CO2 isotope-exchange reactions at high collision energies. This is done by sending two molecular beams hurtling towards each other and analyzing the reaction products. The apparatus in Tim Minton's lab at Montana State University looks kind of like this:
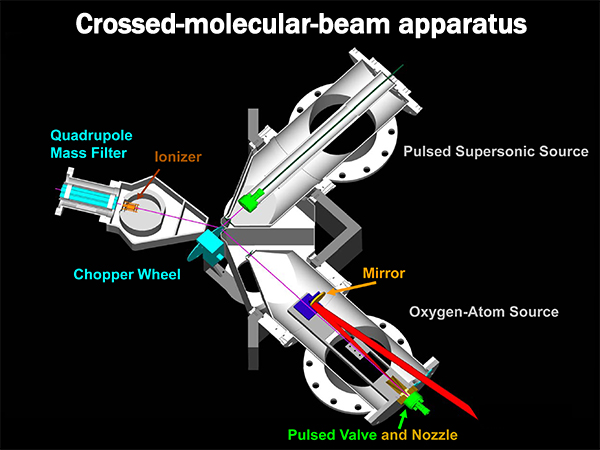
A hyperthermal (~8000 m s-1) beam of O(3P) atoms and O2 molecules is generated by high-power laser heating in the bottom right chamber and a narrow range of velocities is selected by the chopper wheel. A beam of 12C18O2 is introduced via the top right chamber. They interact in the middle (near the chopper wheel) and analyzed with a rotatable mass spectrometer, pictured on the left above. Because these molecules have to fly about 1 meter without colliding with anything else, the whole apparatus has to be at high vaccum (<10-7 Torr, or less than a billionth of an atmosphere!) So, perhaps ironically, the apparatus takes up a whole room with its pumps, forevacuum chambers, and such. Here are some more pictures of it "in action."


All of the distances are fixed and characterized, and the reactant velocities are measured precisely. We go through all this trouble so we can calculate the energy distribution in the products: By measuring the time it takes for a product to arrive at the detector, we know its velocity because we know the flight distance. Therefore, we can derive the translational energy (Etranslation)of the products. Conservation of energy requires that the initial and final energies have the same total energy (Etotal), so the remainder is the internal energy (Einternal):
- Etotal = Etranslation + Einternal
- Key Findings:
- O2 reacts with CO2 at collision energies near 150 kcal mol-1. They exchange oxygen isotopes via a CO4 intermediate. Plus, this reaction can occur adiabatically! It seems as though they are the result of head-on collisions because the products have a lot of internal energy and are scattered opposite the reactants' trajectories. Link to JACS article
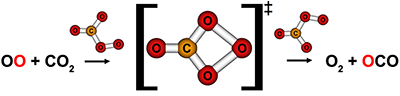
- O(3P) + CO2 collisions near 100 kcal mol-1 yield oxygen isotope exchange and oxygen-atom abstraction. Both reactions probably occur through a CO3 intermediate in that energy range, but they are both short-lived relative to molecular rotation. Still, O(3P) + CO2 isotope exchange may occur as low as the stratosphere because its barrier is only ~25 kcal mol-1; O(3P) atoms with this velocity can be generated when ozone is photolyzed in the UV region. Link to JPCA article
- O2 reacts with CO2 at collision energies near 150 kcal mol-1. They exchange oxygen isotopes via a CO4 intermediate. Plus, this reaction can occur adiabatically! It seems as though they are the result of head-on collisions because the products have a lot of internal energy and are scattered opposite the reactants' trajectories. Link to JACS article
Image: Amazon river plume in the tropical North Atlantic, taken by MODIS/NASA.
Effects of tropical rivers on the oceans
Rivers send 1016 liters (that's 10 million billion liters!) of freshwater into the oceans every year; carried along with that freshwater are minerals, chemical nutrients, and other terrestrial materials that affect the biology and chemistry of oceans. The tropical oceans are particularly starved of nutrients, so rivers have a large impact on their ecosystems.
The Amazon river, the world's largest, discharges fresh water thousands of kilometers into the tropical North Atlantic. Usually, these waters are crystal-clear because there is so little biological activity. When the Amazon river's plume reaches its maximum extent, however, the resulting footprint is visible from space! These plume waters, far from the river mouth, support an unexpectedly efficient biological "pump" that reverses the tropical North Atlanic's normal condition; instead of outgassing CO2 and being a net carbon source to the atmosphere, the area influenced by the Amazon plume becomes a net carbon sink.
Working together with scientists at the University of Georgia, the University of Southern California, San Francisco State University, among others, I am studying the relationship between photosynthesis, carbon sequestration, and phytoplankton community structure as part of the ANACONDAS expedition. The team has hypothesized that this efficient biological "pump" is driven by an unusual assemblage of diatoms and nitrogen-fixing (diazotrophic) cyanobacteria.
- Key Findings:
- We have found that the peculiar association of Richelia intracellularis, a nitrogen fixing organism, with the diatom Hemiaulus hauckii (pictured below; image by D. Caron) can increase the efficiency of carbon export at least twofold in the plume. They account for at least half (and sometimes more than 70%) of the carbon export when they "bloom". These results demonstrate the profound impact that patchy blooms of these species can have on the carbon balance of the Amazon river plume, although it's still not clear under what conditions exactly that these phytoplankton will start blooming. Hemiaulus and Richelia are found nearly everywhere in the oceans, however, so there is a good chance that they play a larger role in both the nitrogen and carbon budgets of the ocean than previously recognized. Link to GRL article
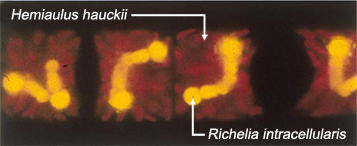
- We have found that the peculiar association of Richelia intracellularis, a nitrogen fixing organism, with the diatom Hemiaulus hauckii (pictured below; image by D. Caron) can increase the efficiency of carbon export at least twofold in the plume. They account for at least half (and sometimes more than 70%) of the carbon export when they "bloom". These results demonstrate the profound impact that patchy blooms of these species can have on the carbon balance of the Amazon river plume, although it's still not clear under what conditions exactly that these phytoplankton will start blooming. Hemiaulus and Richelia are found nearly everywhere in the oceans, however, so there is a good chance that they play a larger role in both the nitrogen and carbon budgets of the ocean than previously recognized. Link to GRL article
- Check out our video abstract! Originally made for the Ocean180 Video Challenge in collaboration with Meg Rosenburg, it was viewed by over 30,000 middle-school students around the world. We won 2nd place, too!
Collaborators: Will Berelson, Ed Carpenter, Victoria Coles, Joe Montoya, Masha Prokopenko, Tish Yager, and Ed Young.
Funding: NSF OCE-093409 (PI Tish Yager), OCE-0961207 (PI Will Berelson), OCE-0961221 (PI Ed Young)



